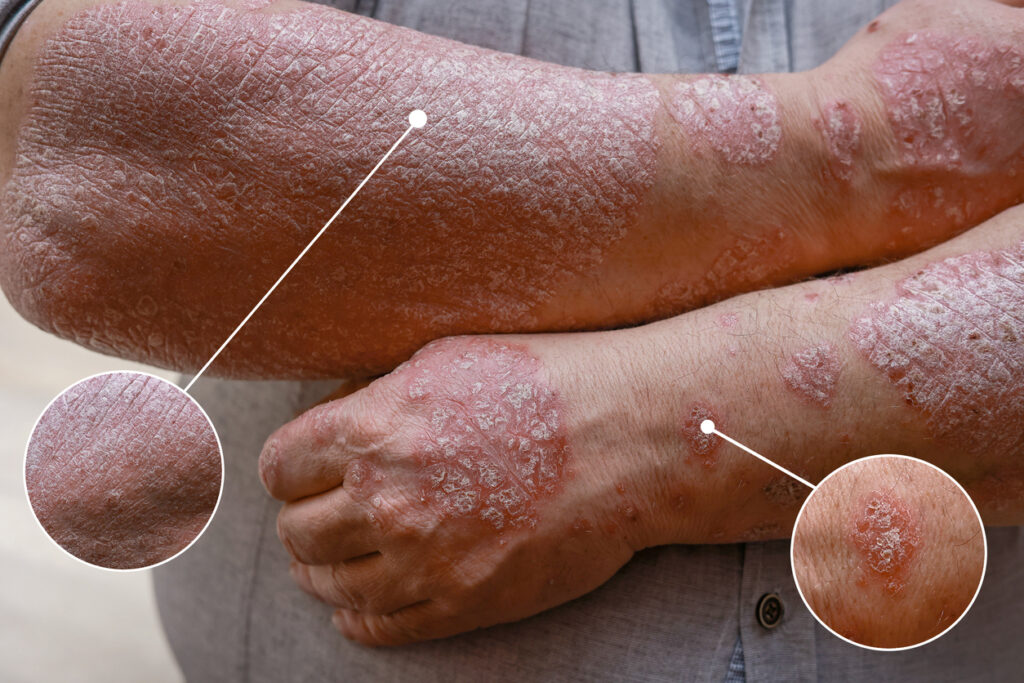
The U.S. Food and Drug Administration (FDA) has approved the Biologics License Application (BLA) for PYZCHIVA (ustekinumab-ttwe) subcutaneous injection and intravenous infusion as a biosimilar to Stelara (ustekinumab).
PYZCHIVA has been approved for the treatment of moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy, active psoriatic arthritis, moderately to severely active Crohn’s disease, and moderately to severely active ulcerative colitis.
Additionally, a provisional determination was granted for PYZCHIVA’s interchangeability.
The FDA’s approval of PYZCHIVA is based on a totality of evidence including analytical, non-clinical and clinical data demonstrating biosimilarity to Stelara, with no clinically meaningful differences in terms of safety, purity and potency. The randomized, double-blind, three-arm, parallel-group, single-dose Phase 1 clinical study (NCT04772274) demonstrated pharmacokinetics (PK) equivalence and comparable safety, tolerability, immunogenicity profiles between PYZCHIVA (SB17) and Stelara in healthy volunteers. The randomized, double-blind, multicenter Phase 3 clinical study (NCT04967508), conducted in patients with moderate to severe plaque psoriasis, demonstrated biosimilarity of SB17 with Stelara through equivalent efficacy and comparable safety and PK profiles up to Week 28 and these primary results were published in the Journal of the American Academy of Dermatology. In addition, the Phase 3 study demonstrated biosimilarity of SB17 to Stelara up to week 28 in terms of efficacy, safety, pharmacokinetics, and immunogenicity, and these data were presented at the 2024 American Academy of Dermatology (AAD) Annual Meeting in March 2024.
PYZCHIVA, developed by Samsung Bioepis, will be commercialized by Sandoz in the United States. Samsung Bioepis and Sandoz entered into a commercialization agreement for SB17 (PYZCHIVA) in September 2023 for the US, Canada, European Economic Area (EEA), Switzerland, and United Kingdom (UK). In the US, the license period for PYZCHIVA will begin on February 22, 2025, according to the settlement and license agreement between Samsung Bioepis and Janssen Biotech Inc.
Samsung Bioepis has a total of 11 biosimilars in its products and pipeline portfolio across immunology, oncology, ophthalmology, hematology, nephrology, and endocrinology. Samsung Bioepis has seven biosimilars approved in the US and four commercially available as of June 2024.