Putting the Squeeze on Melanoma Cells May Make Them More Aggressive
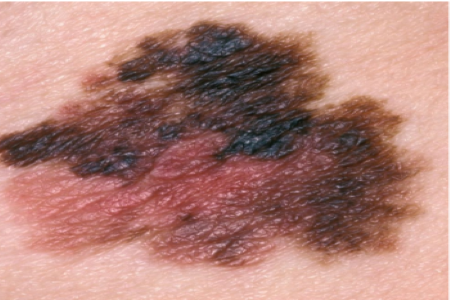
When narrow blood vessels place mechanical pressure on melanoma cells, the cells can become more aggressive, a new study suggests.

When narrow blood vessels place mechanical pressure on melanoma cells, the cells can become more aggressive, a new study suggests.