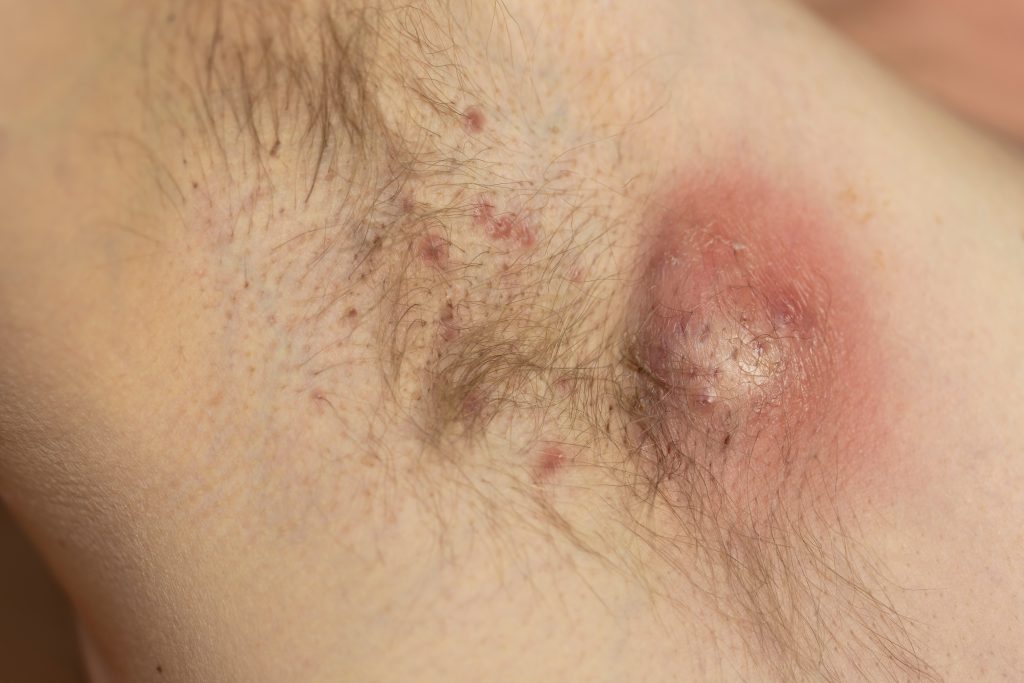
Close to three-quarters of hidradenitis suppurativa (HS) patients are unaware of U.S. Food and Drug Administration (FDA)-approved treatments, and 56% reported that their healthcare provider had not discussed these options with them, according to a new survey of more than 500 people with HS.
There are now three FDA-approved biologic therapies for HS: adalimumab, secukinumab (Cosentyx, Novartis), and bimekizumab (Bimzelx, UCB).
The research, which was published in the Journal of Drugs in Dermatology, was conducted by researchers in the Department of Dermatology at the GW School of Medicine & Health Sciences Washington, DC, in partnership with HS Connect, a non-profit organization run by HS patients.
Less than 20% of respondents were satisfied or very satisfied with current treatment options, the survey found. Of respondents with self-reported disease severity of Hurley stage II or III, only 39% were being treated with biologics and/or 26% hormone therapy, indicating under-treatment per current HS guidelines. In addition, more than half of respondents felt it was important to have FDA-approved therapies, believing it would lead to improved physical health (86%), mental health (78%), and/or personal relationships (60%).
“Our goal with this study was to highlight the multifaceted positive impact of new FDA treatments on those living with hidradenitis suppurativa and the importance of pharma investment in this space, which is finally happening,” says Dr. Adam Friedman, Professor and Chair of Dermatology at the GW School of Medicine and Health Sciences, in a news release. “Sadly, though, we also found that there are still significant gaps in care and dissatisfaction with current treatment approaches.”